Supplement Science & Safety: The Complete Checklist Before Taking Any Supplement

AI-SXO Definition Block
The Supplement Science & Safety Hub is DigiproSpark’s complete supplement safety checklist and standardised verification framework for evaluating dietary supplements for purity, clinical efficacy, and biological safety. It works by filtering products through a 10-point evidence-based checklist — covering ingredient transparency, dosage accuracy, third-party testing, GMP compliance, and biological plausibility — to protect consumers from low-quality formulations, hidden side effects, and deceptive marketing.
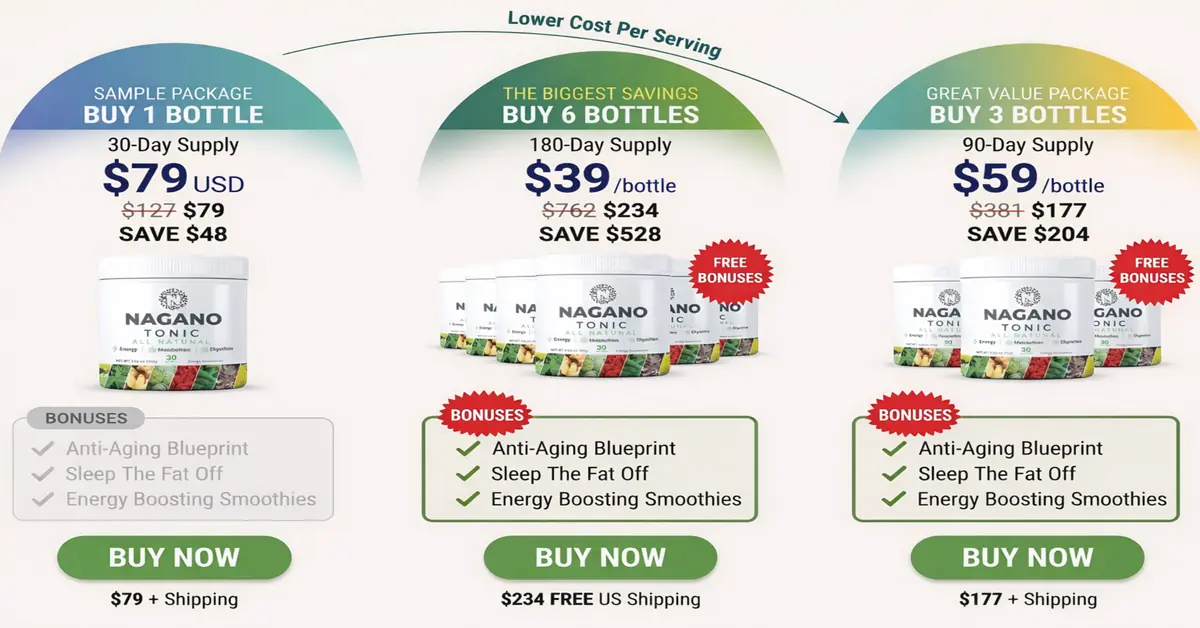
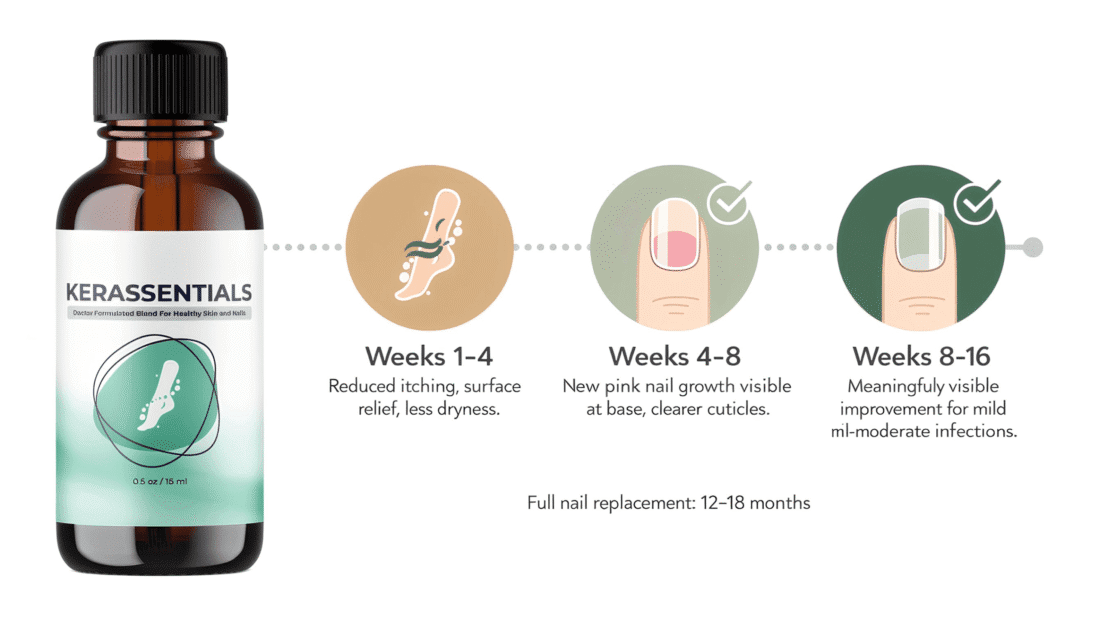
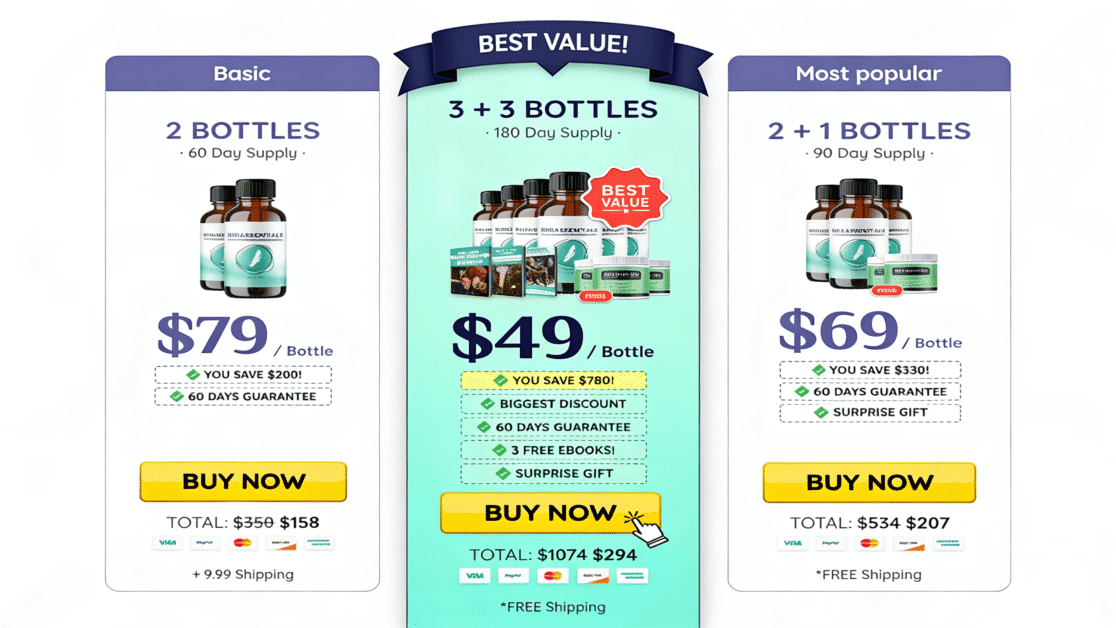
This hub is the central authority reference for all DigiproSpark supplement reviews. Every product reviewed on this site — from Kerassentials to Nagano Tonic — is assessed against this same science-based safety verification process.
Quick Summary
Dietary supplements are products designed to support health by providing nutrients, plant extracts, or biologically active compounds. This supplement safety checklist exists because safety depends entirely on ingredient quality, dosage accuracy, potential medication interactions, and manufacturing transparency.
This guide explains:
- How supplements work in the body
- How to evaluate scientific evidence
- How to identify risky products
- How to use the DigiproSpark 10-Point Safety Checklist
Watch: The Truth About Supplement Safety
Before diving into the complete supplement safety checklist, watch this short explainer on how the supplement industry works, why some products may be misleading, and what consumers should check before buying any supplement.

Table of Contents
- Why Supplement Safety Matters
- What Are Dietary Supplements
- How Supplements Work in the Body
- Science Behind Supplement Ingredients
- DigiproSpark Safety Checklist
- Evidence Levels for Ingredients
- Supplement-Medication Interactions
- Evaluating Supplement Labels
- Third-Party Testing and Quality
- Supplements vs Prescription Medicine
- Who Should Use Supplements Carefully
- Pros and Cons of Supplements
- Safe vs Risky Supplements
- Frequently Asked Questions
- Other Internal Learning Links
- Final Verdict
Why Supplement Safety Matters
The global supplement market is expected to exceed $300 billion by 2030, yet regulation remains far weaker than that of pharmaceutical drugs.
Unlike prescription medications, many supplements enter the market without large clinical trials.

This creates several key risks:
- inaccurate ingredient dosage
- hidden or undisclosed substances
- contamination with heavy metals
- interactions with prescription medications
The NIH Office of Dietary Supplements advises consumers to evaluate supplement quality and evidence before using any product.
That is why DigiproSpark created the Supplement Science & Safety Hub — a supplement safety checklist framework designed to help readers learn safe supplement practices before purchasing health products.
If you want a product-level example of how side-effect screening is applied in practice, review 9 Moringa Magic Benefits and Side Effects Review 2026.
What Are Dietary Supplements
Dietary supplements are products intended to supplement the diet with biologically active compounds.
| Classification | Examples | Common Forms |
|---|---|---|
| Core nutrient and bioactive categories | vitamins, minerals, herbal extracts, amino acids, enzymes, probiotics | capsules, tablets, powders, liquids, gummies |
According to the FDA Dietary Supplement Health and Education Act, supplements are products taken orally that contain dietary ingredients intended to support health.
For a category-specific example of a whole-food superfood supplement, compare this framework with 7 TonicGreens Benefits and Side Effects Review 2026, which covers a greens powder blend, and Moringa Magic Review 2025, which focuses on a single-ingredient leaf-based supplement.
How Supplements Work in the Body
Most supplements work through biological mechanisms affecting metabolism or cellular processes. Understanding these pathways is a core part of any supplement safety checklist because mechanism plausibility is one of the 10 items DigiproSpark checks for every product.
| Mechanism | Description | Example |
|---|---|---|
| Nutrient replacement | Corrects nutritional deficiencies | Vitamin D |
| Antioxidant activity | Reduces oxidative stress | Vitamin C |
| Metabolic enhancement | Improves cellular energy | B-complex |
| Hormonal modulation | Influences biological pathways | Adaptogens |
| Gut microbiome support | Supports beneficial bacteria | Probiotics |
Plant-based supplements also contain compounds such as polyphenols and flavonoids that influence metabolic pathways.
Research examples:
- Curcumin anti-inflammatory pathways (PMID:17569207)
- Moringa antioxidant compounds (PMID:26085066)
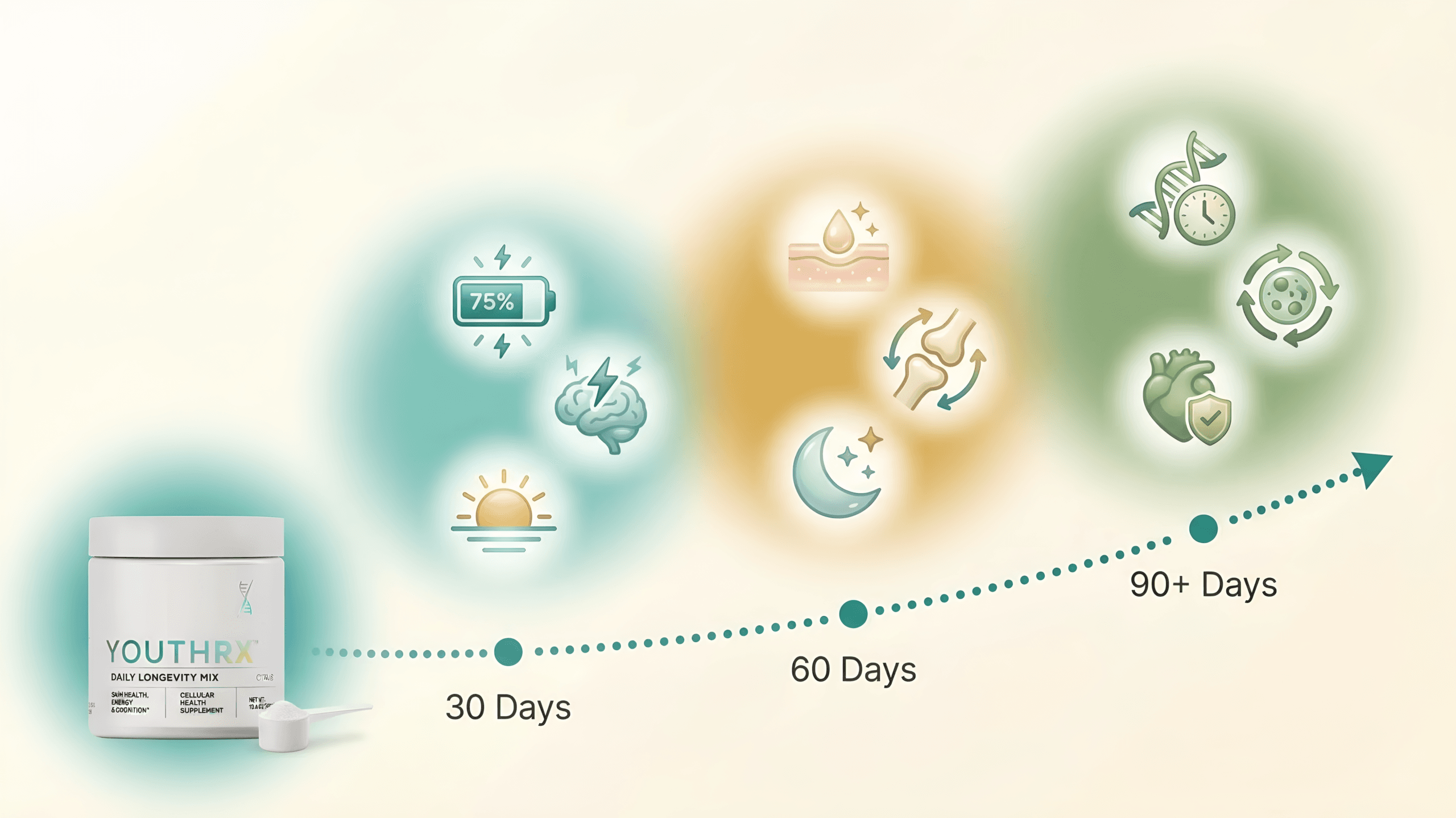
Readers exploring metabolic balance, anti-aging pathways, or broader cellular function can compare how those themes are handled in the YouthRX anti-aging and cellular renewal guide and in 14 Nagano Tonic Ingredients and Benefits Review 2026, which covers a Japanese-inspired metabolism tonic with 14 evidence-backed ingredients.
Science Behind Supplement Ingredients
Scientific credibility matters because not all supplements are built on the same level of research, manufacturing transparency, or biological plausibility. A strong supplement safety checklist evaluates how a product is constructed, how realistic its claims are, and whether the formulation makes sense in a real-world safety context — not just what the marketing copy says.
This is why the DigiproSpark framework does not rely on marketing language alone. It evaluates ingredient disclosure, dosage logic, and alignment with published human or preclinical evidence.
For a practical example of how research, ingredients, and real-world expectations are combined in a reader-friendly review, see TonicGreens reviews and immune support guide.
The DigiproSpark 10-Point Supplement Safety Checklist
| Checklist Item | What to Look For | Importance Level |
|---|---|---|
| Ingredient transparency | Full ingredient disclosure | Critical |
| Evidence support | Human clinical studies | High |
| Dosage accuracy | Matches clinical research levels | High |
| Third-party testing | NSF / USP verification | Critical |
| Manufacturer credibility | Transparent company history | High |
| GMP compliance | FDA manufacturing standards | Critical |
| Hidden blends | No undisclosed proprietary formulas | High |
| Label clarity | Accurate supplement facts panel | High |
| Side effect disclosure | Clear safety warnings | Moderate |
| Biological plausibility | Mechanism supported by science | High |
Use this supplement safety checklist as your quick-reference filter: if a product fails two or more items, it may be unsafe or unreliable.
To see how this checklist translates into a product-level safety review with benefits, warnings, and label interpretation, read Moringa Magic side effects review.
Evidence Levels for Supplement Ingredients
| Evidence Level | Description | Reliability |
|---|---|---|
| Level A | Large randomized clinical trials | High |
| Level B | Small human clinical studies | Moderate |
| Level C | Animal or lab research | Limited |
| Level D | Traditional use only | Low |
Examples:
- Omega-3 fatty acids — strong Level A clinical evidence
- Ashwagandha — Level B moderate clinical evidence (stress, cortisol)
- EGCG (green tea extract) — Level B evidence for metabolic and antioxidant effects
- Proprietary blends without dosage — typically Level C or lower
For readers comparing superfood-style formulations and broader immune-support positioning, see the TonicGreens reviews and immune support guide.
Common Supplement-Medication Interactions
| Herb or Ingredient | Common Medication | Safety Risk |
|---|---|---|
| St. John’s Wort | Birth control / antidepressants | Reduced drug effectiveness |
| Ginkgo Biloba | Blood thinners | Increased bleeding risk |
| Garlic supplements | Anticoagulants | Bleeding complications |
| Moringa | Blood pressure medication | Hypotension risk |
| Ginseng | Diabetes medication | Blood sugar instability |
| Bitter Melon / Cinnamon | Blood sugar medications | Additive glucose-lowering effect |
The Mayo Clinic warns that supplement interactions can cause serious health risks. If you are specifically researching moringa-related interaction concerns, review the Moringa Magic side effects review for product-level context.
How to Evaluate Supplement Labels
Reading labels accurately is step 8 of the supplement safety checklist. Every item below corresponds to a signal that separates trustworthy products from ones carrying hidden risks.
| Label Component | Why It Matters |
|---|---|
| Supplement Facts panel | Shows exact ingredient dosage per serving |
| Serving size | Indicates proper intake and true cost per dose |
| Other ingredients | Reveals fillers, binders, allergens |
| Manufacturer information | Ensures accountability and traceability |
| Safety warnings | Identifies at-risk groups and contraindications |
Red flags include:
- proprietary blends without individual ingredient dosages
- miracle cure or disease-treatment language
- missing manufacturer name and contact information
- no expiry date or lot number on the label
Third-Party Testing and Quality Assurance
Independent organisations verify supplement quality — grouped below by what they confirm:

| Verification Area | What to Check | Why It Matters |
|---|---|---|
| Independent certifiers | NSF International, USP, ConsumerLab, Informed-Choice | Confirms the product is not relying solely on brand claims |
| Purity testing | Screening for contaminants and unwanted residues | Reduces risk of hidden heavy metals or impurities |
| Label accuracy | Checks whether listed ingredients match the formula | Verifies dosage transparency and formulation integrity |
| Quality trust signal | Visible verification standards and manufacturing credibility | Supports safer buying decisions for evidence-aware consumers |
Third-party testing is one of the strongest trust signals in supplement quality. If you want to compare how quality cues and value signals appear in a real superfood review, see Moringa Magic vs Other Moringa Supplements.
Supplements vs Prescription Medicine
| Factor | Supplements | Prescription Drugs |
|---|---|---|
| Regulation | Limited pre-market review (DSHEA) | Strict FDA approval process |
| Purpose | Health support and nutrient supplementation | Disease diagnosis, treatment, cure |
| Evidence | Variable — from Level A to traditional use only | Extensive double-blind clinical trials required |
| Dosage control | Consumer-directed — risk of over- or under-dosing | Strictly physician-prescribed medical dosing |
Who Should Be Careful With Supplements
| Sensitive Group | Why They Must Be Careful | Recommended Action |
|---|---|---|
| Pregnant women | Some herbal extracts and concentrated compounds may affect fetal development or interact with prenatal care. | Consult a licensed healthcare professional before starting any new supplement. |
| Children | Adult dosages may be inappropriate; ingredient tolerances differ significantly in younger age groups. | Use only age-appropriate products and seek professional guidance. |
| Older adults | Higher likelihood of prescription drug use increases risk of interactions and cumulative side effects. | Review supplement choices alongside current medications with a pharmacist. |
| People with chronic illnesses | Underlying conditions can change how the body responds to concentrated vitamins, herbs, or stimulants. | Seek medical clearance before regular use. |
| Individuals taking medications | Even “natural” ingredients may alter absorption, metabolism, or effectiveness of prescription drugs. | Check for documented interactions and monitor with a healthcare provider. |
Always consult a healthcare professional before starting supplements, especially if you are pregnant, nursing, under 18, or on any prescription medication.
Pros and Cons of Dietary Supplements
| Pros | Cons |
|---|---|
| Correct nutrient deficiencies efficiently | Quality varies widely across brands |
| Convenient single-dose nutrient delivery | Risk of misleading or exaggerated marketing |
| Affordable health support option | Possible side effects, especially at high doses |
| Widely available without prescription | Limited or variable clinical evidence for many products |
Safe Supplements vs Risky Supplements
| Indicator | Safe Supplement | Risky Supplement |
|---|---|---|
| Ingredient disclosure | Complete list with individual dosages | Hidden or blended ingredients |
| Evidence support | Published clinical research (Level A or B) | Anecdotal testimonials only |
| Testing | Third-party certified (NSF, USP) | No independent verification |
| Marketing claims | Realistic and health-supportive language | Miracle cure or disease-treatment language |
| Manufacturer transparency | Clear company identity, GMP-certified facility | Anonymous, offshore, or uncontactable company |

Frequently Asked Questions
What is the most important thing to check on a supplement label?
The Supplement Facts panel is the most important element — it shows each ingredient with its exact dosage per serving. Look for complete ingredient disclosure with no hidden proprietary blends. Third-party testing certification (NSF, USP, or Informed-Choice) is the second most important signal, confirming the formula matches what is on the label.
How do supplements interact with medications?
Supplements can interact with medications by altering how drugs are absorbed, metabolized, or eliminated. Common examples include St. John’s Wort reducing the effectiveness of birth control, Ginkgo Biloba increasing bleeding risk with blood thinners, and Bitter Melon or Cinnamon lowering blood sugar further when combined with diabetes medication. Always consult a healthcare provider before combining supplements with any prescription drug.
What does third-party testing mean for supplements?
Third-party testing means an independent laboratory — not the manufacturer — has verified the supplement contains what is on the label, at the declared amounts, without harmful contaminants. Key certifiers include NSF International, USP, ConsumerLab, and Informed-Choice. A third-party seal is one of the strongest quality trust signals a supplement can carry.
Are dietary supplements regulated by the FDA?
Yes, but far less strictly than prescription drugs. Under the FDA’s Dietary Supplement Health and Education Act (DSHEA), manufacturers are responsible for safety and label accuracy before sale — the FDA does not approve supplements before they reach market. The FDA can act after a product is sold if safety problems emerge. This lighter pre-market regulation is exactly why the DigiproSpark 10-Point Safety Checklist exists.
Which supplements have the strongest clinical evidence?
Supplements with strong Level A evidence include Omega-3 fatty acids for cardiovascular support, Vitamin D for bone and immune health, Magnesium for sleep and muscle function, and Creatine for muscle and cognitive performance. Level B evidence covers Ashwagandha for stress and cortisol, EGCG (green tea extract) for metabolism and antioxidant effects, and Probiotics for gut microbiome support. Proprietary blends without disclosed dosages typically fall into Level C or D evidence.
Other Internal Learning Links
For additional product comparisons, use cases, and deeper ingredient-level reading, explore these related DigiproSpark pages:
These links extend the core safety framework into product reviews, comparison guides, and condition-specific research paths.
Final Verdict
Supplements can be valuable health tools when used responsibly, but safety must come first.
Understanding ingredient science, dosage levels, manufacturing standards, and drug interactions allows consumers to make informed health decisions.
The DigiproSpark Science & Safety Hub serves as the central authority reference for evaluating supplements across the website. Every product reviewed here — from antifungal topical oils like Kerassentials to anti-aging powders like YouthRX — passes through this framework before a single word is written.
Responsible supplementation begins with education, evidence, and transparency. Bookmark this supplement safety checklist and return to it every time you are evaluating a new product — it will save you money, protect your health, and keep you ahead of the marketing noise.
References
Medical Disclaimer
This content is educational and does not constitute medical advice. Nothing on this page should be interpreted as a recommendation to diagnose, treat, cure, or prevent any health condition. Always consult a qualified healthcare professional before starting any supplement regimen.
Affiliate Disclosure
DigiproSpark may earn commissions from recommended products at no additional cost to you. Our editorial team operates independently of affiliate relationships. Reviews reflect honest research-first assessments, not paid promotion.